A healthcare professional in Brazil is under investigation after being accused of selling prescription-only weight-loss medication without authorisation. The case, reported in the coastal city of Santos, São Paulo, highlights growing anxieties around the misuse of anti-obesity drugs within clinical environments.
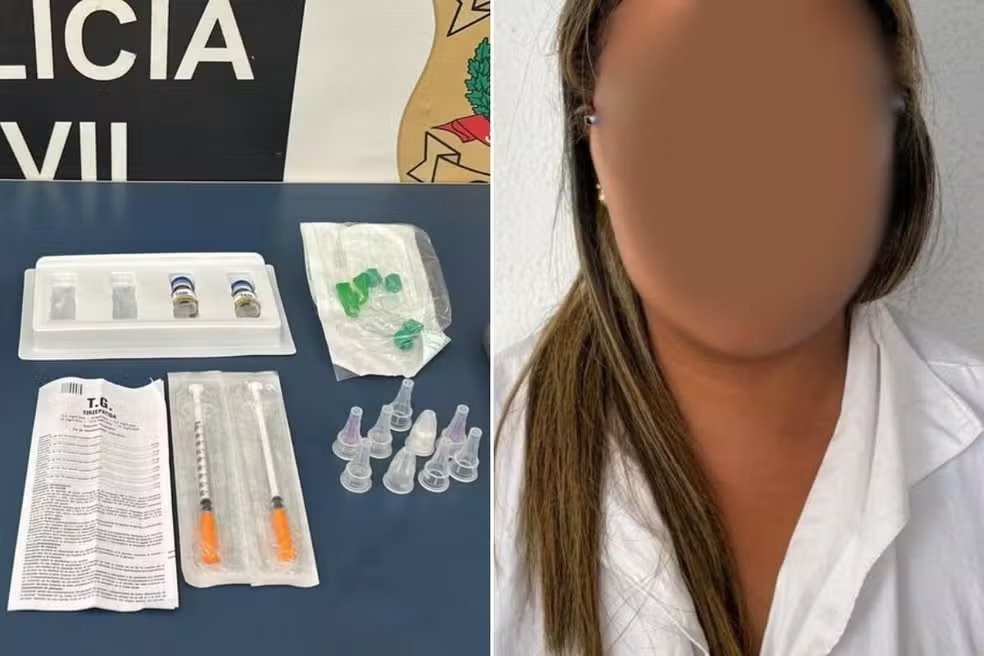
According to Brazilian civil authorities, a 35-year-old nursing technician was taken for questioning following a search-and-seizure operation. Police initiated inquiries after receiving information suggesting that the professional and a colleague were storing and commercially distributing tirzepatide – marketed internationally as Mounjaro – without legal approval. Search warrants were issued for multiple locations, including the hospital where she was employed and the homes of both individuals.
During the operation, investigators located two injectable ampoules reportedly acquired without medical prescription. Local law enforcement stated that the medicine had been purchased online for approximately R$1,000 and delivered by post. The woman was released following her testimony, but her mobile phone and the confiscated medication were seized to support ongoing inquiries. The incident was logged under offences linked to unauthorised medication sales and regulatory violations.
The healthcare institution involved, a hospital in Santos, confirmed that both employees were suspended pending the outcome of the investigation. In an official statement, the hospital emphasised that it was cooperating fully with Brazilian authorities and clarified that it was not directly implicated in the alleged activities.
Growing global scrutiny over appetite-regulating injections
Medications belonging to the incretin class, such as tirzepatide, semaglutide (known commercially as Ozempic and Wegovy), have gained widespread public attention for their effects on weight reduction. These drugs mimic hormones involved in appetite regulation, prolonging satiety and reducing food cravings. Clinical research, including studies published in The New England Journal of Medicine, has demonstrated significant weight-loss outcomes in patients undergoing medically supervised treatment.
However, scientists and regulators worldwide have raised concerns over improper usage, counterfeit products and unlicensed distribution. Health agencies in multiple countries caution that unsupervised consumption may increase risks such as gastrointestinal complications, hypoglycaemia and inappropriate dosing.
This latest case in Brazil adds to growing international debate surrounding ethical prescribing practices and access control for medications designed primarily for diabetes management but increasingly used for aesthetic weight loss. Regulatory bodies continue to stress that such treatments should only be administered with professional guidance and proper diagnostic screening.
Authorities in São Paulo are expected to release further updates as the Brazilian inquiry progresses. The case reinforces the necessity for robust oversight in hospital settings to safeguard patients and ensure safe, evidence-based use of emerging pharmaceutical therapies.